Vaccination in Poland: how to apply and other things you need to know
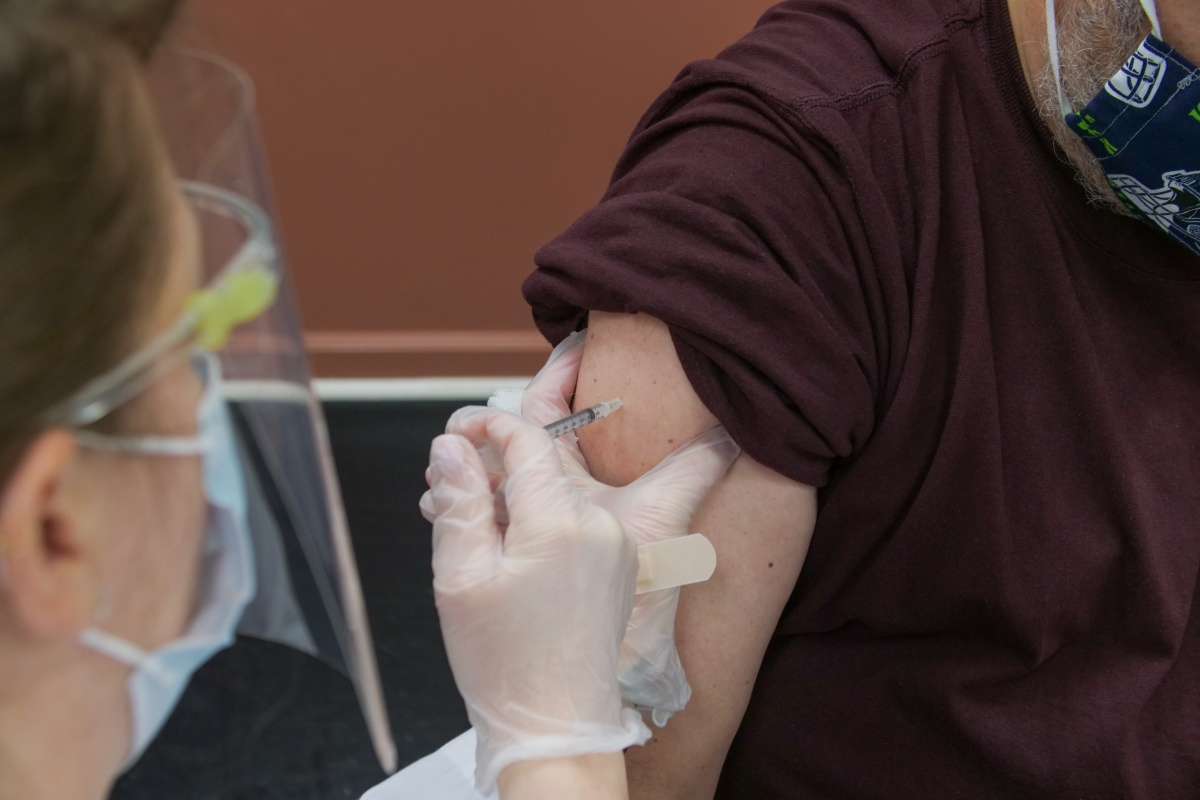
The vaccination is free and voluntary. Over a month ago, the Polish government signed contracts for the purchase of COVID-19 vaccine with three companies: Pfizer BioNtech, Johnson & Johnson, and AstraZeneca. On December 27, 2020 the wide distribution of vaccines in Poland has begun. In this article, we gather important things you need to know about vaccination in Poland.
How to apply for vaccination?
There are two ways to apply, which are registration by phone or by online form. However, if you are a healthy young adult, you might need to wait a little bit longer as the vaccine distribution in Poland is still in stage 1. Vaccination in stage 1 belongs to the elderly aged over 60 years old.
How is the process of vaccination going?
Vaccinations in Poland are carried out in these stages:
- Stage 0 (from December 27, 2020)
The vaccines belong to the people mentioned in this stage, such as; healthcare worker (including individual practitioner), employees of social welfare homes and employees of municipal social welfare centers, as well as auxiliary and administrative staff in medical facilities, including sanitary stations - epidemiological, parents of premature babies, and retired doctors with the right to practice.
- Stage 1 (from January 25, 2021)
Residents of social welfare homes, care and treatment facilities, nursing and care facilities, and other places of stationary stay. People over 60 years old in order from the oldest, uniformed services, including the Polish Army, and teachers.
- Stage 2 (to be announced)
People under 60 years old with chronic diseases that increase the risk of severe COVID-19, or during diagnostics and treatment requiring repeated or continuous contact with health care facilities, people directly ensuring the functioning of the state's basic activities and at risk of infection due to frequent social contacts.
- Stage 3 (to be announced)
General vaccination of the rest of the adult population will be implemented.
Vaccine reaction
Vaccination adverse events can be mild, serious, or severe. In Poland, the adverse vaccine reactions are recorded on average once in 10,000 cases. The most common reaction reported are redness and short-term shortness at the injection site.
Read also: Who can cross the Polish border without a 10-days quarantine?
--
Source: gov.pl/web/szczepimysie

